Anti-amyloid therapies in Alzheimer’s disease: clinical evidence, ARIA management, and real-world implementation

A new era in the treatment of Alzheimer’s disease (AD) has begun in Switzerland. With the approval of the anti-amyloid monoclonal antibody donanemab by Swissmedic, the first disease-modifying therapy for Alzheimer’s disease is now available. For the first time, treatment is no longer limited to symptomatic relief, but aims to slow the underlying disease process itself.
Is the healthcare system ready for disease-modifying Alzheimer’s therapies?
Implementing anti-amyloid therapies brings significant clinical and organizational challenges. Their use requires careful patient selection, structured infrastructure, and close monitoring to detect and manage potential side effects such as amyloid-related imaging abnormalities (ARIA).
At the 2025 last November, Dr. Gil Rabinovici (University of California, San Francisco) shared insights from the U.S. experience with amyloid-targeting therapies. He emphasized that the rollout has been relatively slow, even in experienced centers. By the end of October 2025, only a limited number of patients with Alzheimer’s disease (n = 163) had been treated with anti-amyloid therapy at his center, reflecting the complexity of real-world implementation. Importantly, he noted that careful patient selection and structured monitoring protocols are essential to reducing the risk and severity of ARIA.
While awaiting approval in Switzerland, memory centers proactively prepared for safe and effective implementation. As part of our , organized in collaboration with the , we invited two experts to discuss practical implementation strategies, early clinical experiences, and critical safety considerations surrounding anti-amyloid therapies.
Anti-amyloid therapies: practical insights and first experiences
In the first webinar, Dr. Aurélien Lathuilière from the Memory Center at the Geneva University Hospitals (HUG) focused on translating clinical trial evidence on anti-amyloid therapies (AATs) into routine clinical practice.
He structured his presentation in four parts: (1) reviewing the scientific evidence underlying anti-amyloid therapies; (2) exploring how trial results can be translated into routine clinical care; (3) identifying key bottlenecks and strategies for risk management; and (4) sharing practical insights from the implementation planning process in Geneva.
Clinical evidence: key trials
To start, Aurélien guided the audience through the two pivotal phase III trials that form the foundation for current clinical practice:

Patient population:
- Predominantly patients in their 70s with early symptomatic AD.
- APOE ε4 carriers represented a significant portion of the study population (around 70%).
Efficacy:
- Both drugs reduced cognitive decline by approximately 29–33% over 18 months compared with placebo.
- Clinical effects were supported by robust reductions in amyloid plaque burden confirmed by PET imaging.
Safety considerations: ARIA
- The most significant adverse events include edema/effusion (ARIA-E) and microhemorrhages/superficial siderosis (ARIA-H).
- Risk is highest in APOE ε4 homozygotes and during the first six months of treatment.
- Other risk modifiers include vascular pathology, microbleeds, and uncontrolled hypertension.
Moving from clinical trials to real-world implementation
Aurélien emphasized that moving from clinical trials to real-world implementation is a multifaceted challenge. Patients seen in memory clinics differ from trial participants: they are often older, have multiple comorbidities, take several medications, and may not have immediate access to biomarker testing or intensive MRI monitoring. To address these challenges, Swiss Memory Clinics are focusing on four essential pillars:
- Structured patient selection
- Accurate clinical stage and comorbidity assessment
- Confirmation of biomarkers
- Standardized MRI assessment
- Baseline imaging to evaluate eligibility
- Follow-up MRIs to detect ARIA early
- Patient counseling and shared decision-making
- Clear communication of expected benefits (~30% reduction in cognitive decline over 18 months) and risks, especially ARIA.
- Multidisciplinary coordination
- Collaboration between dementia specialists, neuropsychologists, radiologists, nurses, and administrative staff.
Bottlenecks in real-world implementation
“The major challenges of anti-amyloid therapies are organizational rather than pharmacological,” Aurélien emphasized. The drugs work as designed, but the healthcare system must be prepared to deliver them safely and efficiently.
Key bottlenecks memory clinics must anticipate:
- Patient selection
- MRI capacity
- Safety monitoring
- Human resources
To support early implementation, Aurélien referred to several recently published practical guidance papers:
These recommendations translate strict trial inclusion and exclusion criteria into practical guidance for everyday care. For instance, in Switzerland it was considered safer to exclude patients with cerebral amyloid angiopathy (CAA). The goal is not to treat as many patients as possible, but to treat the right patients under conditions that preserve benefits and minimize risk. The practical resources also include recommendations from the Swiss Society of Neuroradiology on MRI sequences. Aurélien highlighted the following practical steps:
- Estimate the number of potential candidates (approximately 5–15% of AD cases).
- Assess referral pathways and waiting times; long waits may cause the optimal treatment window to close.
- Establish a multidisciplinary board to review eligibility and support clinical decision-making.
- Implement a biomarker-based diagnostic pathway.
- Clarify access to APOE genotyping.
- Clarify access to MRI and ensure standardized sequences and reporting.
- Train colleagues in emergency medicine, stroke care, and primary
The Geneva experience: Planning before approval
Even before final regulatory decisions in Switzerland, the Memory Center at Geneva University Hospitals (HUG) established a multidisciplinary task force to prepare for potential implementation. First, they estimated the eligible population using a well-established data registry. Second, the task force developed internal Good Clinical Practice (GCP) materials, such as patient information brochures, consent forms, eligibility checklists, MRI checklists, and ARIA management algorithms tailored to their hospital.
Aurélien concluded by presenting early experiences with lecanemab at a specialty memory clinic in Washington, highlighting two severe ARIA cases (Paczynski et al., JAMA Neurology, 2025). He emphasized that structured infrastructure, careful patient selection, and a gradual rollout are key. Experience over time will help refine clinical practice, underscoring the importance of starting slowly and adapting as real-world data accumulate.
ARIA – prediction, detection and management
In the second webinar, PD Dr. med. Piotr Radojewski (Translational Imaging Center Bern and University Institute of Diagnostic and Interventional Neuroradiology, Inselspital, University Hospital Bern) offered a deep dive into the amyloid-related imaging abnormalities (ARIA), its detection and management.
Understanding ARIA: Pathophysiology and imaging
ARIA is thought to result from the brain’s response to rapid removal of amyloid plaques by anti-amyloid therapies. A leading hypothesis is that accelerated plaque clearance overwhelms perivascular pathways, causing transient blood–brain barrier dysfunction. As a result, fluid and inflammatory cells can leak into brain tissue, leading to localized edema and sulcal effusions. These changes are detectable on MRI using FLAIR sequences and are classified as ARIA-E (edema/effusion).
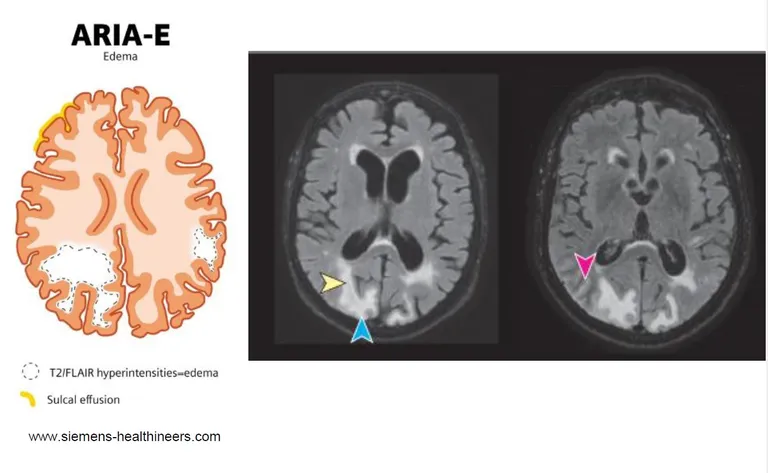
In parallel, compromised vessel integrity can allow small amounts of blood to escape into surrounding tissue. Iron-sensitive MRI sequences, such as T2* or susceptibility-weighted imaging (SWI), reveal microhemorrhages and linear superficial siderosis, collectively classified as ARIA-H (hemorrhage).
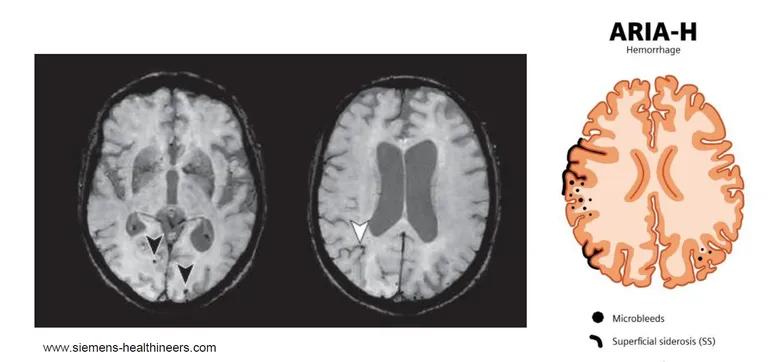
Historically, ARIA was first reported in early trials around 2009 and later categorized into E and H subtypes. Early findings showed that ARIA can be severe, but many cases are asymptomatic and resolve on follow-up imaging—underscoring the transient nature of ARIA-E and the importance of routine monitoring.
Lessons from trials
Piotr emphasized that clinical trials have been instrumental in shaping our understanding of ARIA. Although the clinical implementation of aducanumab ultimately failed and the drug was withdrawn from the market, its development program provided critical insights into the safety profile of anti-amyloid therapies as a class.
The ARIA-E severity grading system—mild, moderate, or severe based on lesion size—was established during early trials and remains valid today. ARIA-E and ARIA-H can occur simultaneously. While ARIA-E typically has a transient course and resolves on follow-up imaging, ARIA-H findings (microbleeds and superficial siderosis) may persist on MRI for prolonged periods.
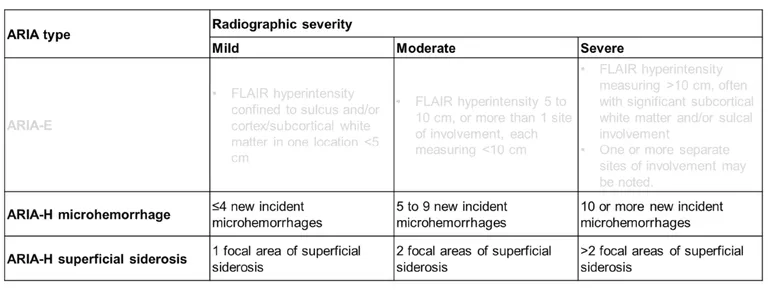
Across subsequent trials with other compounds, including lecanemab and donanemab, ARIA has likewise been observed but has remained predominantly asymptomatic. These findings underscore the necessity of structured, repeated MRI monitoring to detect ARIA early and manage it appropriately.
Predictors of ARIA
Beyond describing ARIA itself, trial data have clarified who is at highest risk. The strongest predictor of ARIA is the treatment itself: ARIA is a mechanism-related effect of amyloid-targeting therapies. Importantly, modified dosing regimens—such as slower titration schedules—can reduce the incidence and severity of ARIA. When ARIA does occur, it typically presents early after treatment initiation, as shown in CLARITY-AD and other phase III programs.
The second most important predictor is APOE genotype:
- APOE ε4 homozygotes carry the highest risk.
- APOE ε4 heterozygotes have an intermediate risk.
- Non-carriers have the lowest risk.
This genetic effect has been consistently observed across trials and is now central to pre-treatment risk assessment and patient counseling.
Barriers to ARIA monitoring in real-world practice
While trial data have clarified ARIA risk factors, the implementation of ARIA monitoring in real-world practice presents a separate set of challenges. To better understand these, the European neurological and neuroradiological societies established a joint committee and conducted a survey among neurologists and neuroradiologists. The goal was to identify potential barriers before large-scale implementation of anti-amyloid therapies. Survey results showed moderate to high confidence among specialists in recognizing and managing ARIA. However, when asked about practical readiness, responses were more cautious.
Key barriers identified:
- Logistics and workflow organization
- Availability of MRI scanners
- Radiological and neuroradiological expertise
Piotr mentioned that preparedness for anti-amyloid therapies is not only about understanding ARIA pathophysiology, it requires much more such as scanner availability, trained staff, defined communication pathways and most importantly standardized protocols and reporting templates.
Take-home message:
- Standardization is essential. European experts recommend using SWI and 3D FLAIR for ARIA detection, documenting individual risk factors in the baseline MRI report, and applying structured ARIA grading with clear severity classification.
- National regulations matter. In Switzerland, the Swissmedic label specifies the use of T2*-weighted imaging and 2D FLAIR. Baseline MRI must be performed within three months prior to treatment initiation, with follow-up imaging required before the 2nd, 3rd, 4th, and 7th infusions, and additionally whenever symptoms suggest ARIA.
- Prompt recognition saves lives. Any new neurological symptoms require immediate MRI and structured reporting. Crucially, treating physicians and emergency staff must be aware if a patient is currently receiving anti-amyloid antibody therapy, as ARIA can mimic acute stroke. Inappropriate thrombolysis or anticoagulation in these patients can be fatal. Awareness in emergency settings must therefore explicitly include knowledge of the ongoing treatment.
- Communication is key. Safe implementation depends on well-defined communication pathways between neurology, neuroradiology, emergency departments, and referring physicians—particularly in a decentralized healthcare system like Switzerland’s.
- Preparedness goes beyond knowledge. Infrastructure, scanner availability, trained staff, standardized protocols, and coordinated triage systems are just as important as understanding ARIA pathophysiology.
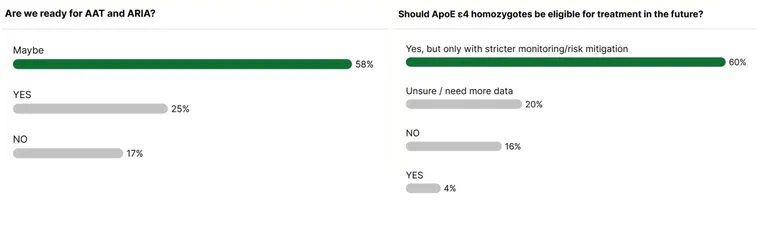
Piotr closed the webinar with a question to the audience: “Are we ready for anti-amyloid therapies and ARIA?”
The responses reflected both progress and caution. A majority (58%) selected “maybe,” acknowledging that while important groundwork has been laid, key structural and logistical challenges remain. Twenty-five percent were optimistic and answered “yes,” expressing confidence in current preparedness. However, 17% felt that Switzerland is not yet ready for widespread implementation.
A second question addressed the eligibility of APOE ε4 homozygotes, the group known to carry the highest risk for ARIA. Most participants agreed that treatment should be available to this high-risk population, but only under stricter monitoring protocols and enhanced safety measures.
These responses capture the current moment well: the field is moving forward, but with careful consideration.