Women, diversity and brain health: New perspectives on dementia risk

Dementia risk arises from a complex interplay of biological and social factors. Recent research highlights how menopause and cardiometabolic health influence women’s brain ageing, while sex/gender, race/ethnicity, and socioeconomic factors intersect to shape Alzheimer’s risk. Insights from and in our underscore the need for integrated approaches to understanding and preventing cognitive decline.
Risk factors for cognitive decline in the context of gender differences
Dr. Louise Schindler from King’s College London spoke in the Dementia Update – Clinical Series (co-hosted with the Swiss Memory Clinics), highlighting the connections between menopause, cardiometabolic health, and women’s brain health. She began the webinar by addressing a common misconception: menopause is often thought of simply as the end of reproductive life, but it represents a much broader physiological and neurological transition. Medically, menopause is defined as the moment a woman has gone 12 consecutive months without a menstrual period which marks the end of reproductive life. The perimenopause is the transition period during which estrogen levels fluctuate dramatically before declining. Women commonly experience symptoms such as hot flushes, fatigue, irritability, mood disturbances, sleep problems, and cognitive complaints, including what is colloquially known as “brain fog.” Interestingly, many, if not most, symptoms are in fact neurological in nature.
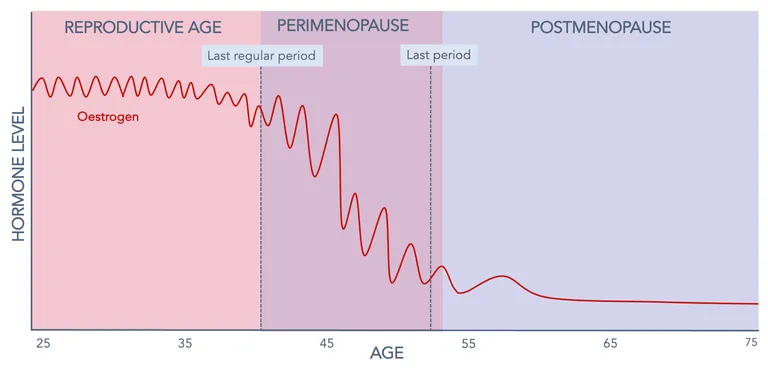
Figure 1: Production of estrogens in women with age.
Estrogen exposure and brain health
To explore whether menopause might be linked to brain health, Louise began with evidence on estrogen exposure. Animal and experimental studies suggest that estrogen has neuroprotective properties, supporting synaptic plasticity, cell survival, neuronal growth, and myelination. This provides a biologically plausible explanation for why hormonal changes during menopause could influence brain health later in life.
In humans, lifetime estrogen exposure has also been linked to brain health. In her first PhD paper, Louise and colleagues studied over 10,000 postmenopausal women from the UK Biobank, using reproductive span, the time between first and last menstrual period, as a proxy for lifetime estrogen exposure (LS Schindler et al. Neuroimage Clin. 2022:36:103239). They found that a longer reproductive span may be associated with healthier brain markers, such as lower white matter hyperintensity volume and a younger-appearing brain age. Other observational studies have reported similar patterns.
However, Louise cautioned that causal evidence is lacking. A recent Mendelian randomization study found no robust causal link between estrogen exposure and brain outcomes, and effect sizes in observational studies are modest.
Menopausal transition and brain health
If lifetime estrogen exposure is not clearly causal, does the menopausal transition itself affect the brain? Animal studies suggest several possible mechanisms: estrogen depletion has been linked to mitochondrial dysfunction, neuroinflammation, and accelerated amyloid accumulation, all processes associated with Alzheimer’s disease. However, direct evidence in humans remains limited. One study compared pre-, peri-, and postmenopausal women, finding differences in brain structure, glucose metabolism, connectivity, and amyloid deposition. Interestingly, some changes appeared to stabilize or even partially recover after menopause, suggesting that the brain may adapt to the new hormonal environment rather than continuing to decline.
Louise emphasized an important methodological challenge in menopause research: it is extremely difficult to disentangle the effects of endocrine aging (menopause) from chronological aging. Postmenopausal women are, by definition, older, and statistical adjustments cannot fully solve this issue. Headlines on this topic should be interpreted with caution, as these methodological nuances are often missing from news coverage.
Menopausal symptoms as indicator of brain health
Beyond hormone levels, Louise addressed whether menopausal symptoms themselves are markers or even contributors to differences in brain health. Depression, anxiety, hot flashes, sleep and brain fog during perimenopause have all been associated with poorer cognitive performance.
Studies by Thurston and colleagues found that vasomotor symptoms (hot flashes and night sweats) were associated with greater white matter hyperintensity volume and amyloid pathology, independent of oestrogen levels. This suggests symptoms may influence brain health beyond hormonal changes and raises an important question: could managing symptoms such as hot flashes have benefits for long-term brain health? While symptom management is primarily aimed at improving quality of life, Louise noted that it might also offer a protective effect for the brain, though more research is needed to confirm this.
Contribution of cardiometabolic health to the menopause brain relationship
One of the main research interests during Louise’s PhD was the link between menopause, cardiometabolic health, and the brain. Studying nearly 10,000 UK Biobank women – approximately one-third premenopausal, two-thirds postmenopausal – she found that postmenopausal women had higher LDL cholesterol, triglycerides, and blood sugar (HbA1c). Longitudinal analyses showed increases in BMI and waist-to-hip ratio, driven by rising waist circumference, consistent with central fat accumulation.

Figure 2: Cardiometabolic health across menopausal years. et al. Front Glob Womens Health. 2023 Dec 21:4:1320640
This matters because cardiometabolic risk factors are robustly linked to brain aging markers, including white matter hyperintensities, lower cortical thickness, and reduced grey matter, and the 2024 Lancet Commission on Dementia Prevention emphasized the contribution of modifiable vascular and cardiometabolic risk factors to dementia risk. Crucially, unlike hormones, cardiometabolic health is modifiable, offering a potential avenue for intervention.
Take-home message:
Louise concluded her presentation by highlighting the key message from her presentation:
- Menopause is a neurological and cardiometabolic transition, not just a reproductive one
- Observational evidence links estrogen exposure to brain health, but the causal evidence is weak
- Menopausal symptoms may flag women at greater brain health risk – treating them could matter
- Menopause drives cardiometabolic changes that are established risk factors for brain health and dementia
- Menopause may be a critical window for brain health research and prevention
An intersectional approach to understanding Alzheimer's disease risk
In the Dementia Update – Virtual Series, Dr. Sivaniya Subramaniapillai from Unisanté, University of Lausanne explored how sex/gender, race/ethnicity, and socioeconomic factors intersect to shape Alzheimer’s risk.
Sivaniya opened with a striking paradox: the populations most affected by Alzheimer’s disease are often underrepresented in the very studies designed to understand it. In the United States, Black and Hispanic/Latino populations show higher dementia risk compared to non-Hispanic white individuals. Globally, over 60% of the global dementia burden falls on individuals in low- and middle-income countries. Approximately two-thirds of Alzheimer’s cases occur in women. Yet, many large neuroimaging datasets are disproportionately composed of White, highly educated participants from high-income settings with access to advanced imaging technologies. Few have systematically accounted for female-specific factors such as menopause, reproductive history, or hormonal exposures. Large portions of the global population still lack access to brain scanners, limiting representation in brain research. As a result, current models of Alzheimer’s disease risk may not fully generalize to the populations bearing the greatest burden.
Sex, gender and social determinants
The terms sex and gender are often conflated, so Sivaniya defined them. Sex refers to biological attributes, including chromosomes, hormones, and physiology, while gender reflects socially constructed roles, norms, and lived experiences. In Alzheimer’s disease, sex plays a critical role. For example, estrogen has neuroprotective effects, influencing synaptic plasticity, glucose metabolism, and neuroinflammation. The menopausal transition, marked by a sharp decline in estradiol, has been proposed as a potential window of increased vulnerability for brain aging in women. In addition, genetic risk factors, such as the APOE ε4 allele, may exert sex-specific effects, with some evidence suggesting a stronger association with Alzheimer’s risk in women than men.
Gender, meanwhile, shapes risk through pathways that are social. These include differences in educational opportunities, caregiving roles, occupational exposures, and access to healthcare. Gender norms may also influence how symptoms are perceived, reported, and acted upon, both by individuals and clinicians.
Considering sex and gender together provides a more complete account of why Alzheimer’s risk and presentation differ across populations.
Intersectionality in action
Intersectionality, a framework coined by Kimberlé Crenshaw, holds that overlapping identities do not simply add up. They interact and produce patterns of risk that are invisible when factors are studied one at a time.
Recent studies are beginning to show what this looks like in practice. Applying this framework to Alzheimer’s disease reveals important insights:
Who goes undetected?
et al. (2024) examined over 3,100 individuals with autopsy-confirmed Alzheimer's pathology and found that nearly 5% had never been diagnosed with MCI or dementia during life. Hispanic individuals, those living alone, and individuals with higher education were more likely to remain undiagnosed, with weaker evidence for a similar pattern in women.
The intersectional findings were notable: the association with education was observed in men but not women, and women with a non-English primary language were more likely to have been diagnosed before death, though the reasons remain unclear, and may relate to differences in cognitive testing or clinical assessment. These results highlight that social determinants of health shape whether Alzheimer's pathology is recognised during life.
Who carries which pathology?
et al. (2025) examined neuroimaging markers of dementia in over 3,400 dementia-free adults from the HABS-HD cohort. They found that the distribution of pathology differed across groups. Black men showed lower amyloid burden but higher levels of cerebral small vessel disease compared to non-Hispanic White participants. Black and Hispanic women exhibited greater tau deposition in the medial temporal lobe compared to their non-Hispanic White counterparts. These findings suggest that the biological profiles associated with dementia risk are not uniform across populations, and that the relationship between amyloid, tau, and vascular pathology may vary by social and demographic context.
Who shows more brain aging, and why?
et al. (2024) used deep learning on fMRI and EEG data from over 5,300 participants across 15 countries to estimate brain-age gaps (the difference between predicted brain age and chronological age). They found larger brain-age gaps in participants from Latin American and Caribbean (LAC) datasets compared to non-LAC datasets, with a clear gradient from healthy controls to MCI to Alzheimer’s disease. Within LAC, females showed larger brain-age gaps than males in both healthy and Alzheimer’s disease groups. At the country level, socioeconomic inequality, pollution, and overall disease burden were associated with larger brain-age gaps. These findings suggest that differences in brain aging are not explained by disease processes alone, but are also shaped by broader social and environmental conditions.
Overcoming research barriers
Sivaniya closed by highlighting three major challenges in advancing inclusive dementia research: recruitment, trust, and analysis. Reaching underrepresented populations requires sustained community engagement, while historical injustices, such as unethical medical studies, continue to shape levels of trust in research. Additionally, studying intersecting identities can limit statistical power, complicating analysis.
Innovative approaches are beginning to address these barriers. For example, portable low-field MRI systems offer a promising way to expand access to neuroimaging and improve representation in research. Sivaniya's own , with collaborators from Unisanté/UNIL, University College London, and Queen Mary University is putting this into practice: using portable MRI in London to recruit from communities typically underrepresented in brain aging research.
Toward more representative brain science
A central takeaway from these talks is that Alzheimer’s disease cannot be fully understood through a purely biological lens. Risk emerges from the interaction between biology, lived experience, and structural conditions.
As Sivaniya noted, “The science of Alzheimer’s disease will only be as precise as the populations included in studies.” Ensuring those populations are truly representative is essential for both equity and scientific accuracy.